In the transformation of the zirconia, several symmetries are reached by a kind of coordenate moving of oxygens (sulattice), depending on the temperature. If only sublattice is taked in consideration, a particular case can be mentioned, that is the martensitic tranformation of the zirconia. From a practical view-point, when the sublattice undergoes further changing, are common changes in the crystal symmetry. As a whole, lattice and sublattice can be change in a major or minor extension, with no coupling in magnitude. In a broad sense, a cation is desordered when occupy two different sites in a crystalline structure, which is determined by the sublattice. From this point, an other concept emerges that is concept of desorder. Such phenomena, migth be more realistic at complex crystalline structure with multiple occupation sites, with distict coordination polyhedra and/or multiplicity of unitary cells. However, one or another or both can be exhibits some kind of small desorder but that can be only a distortion based on slight departure of ion of a proper position at crystalline lattice. At almost simple compounds, both lattice and sublattice follow a long range order. From this point, the complements of question can be approched, the idea of long range order should be considered with some carefull. The sublattice defines the first coordination-sphera of a cation being that both lattice/sublattice are fundamental to derive the space group. By consequence, cations Zn, Co, Fe Nb compose the lattice of these compounds. In this terminology, oxigen ions compose the sublattice of ZnO, CoO Co2O3, Fe2O3, Nb2O5. Ex.: oxygen in the oxides compose the sublattice. The terminology is as follow: in a general way, the chemical element that appears in several compounds that represents or are significant at a class of compounds receives the name sublattice. If a lattice exist is mandatory the existence of a sublattice and vice-versa, mettalic lattice are particular cases. A priori, both concepts lattice and sublattice make sense only at crystal structures. In this sense, when is necessary the following assumptions can be used. Then, a solid concept should be constructed from previous assumptions. 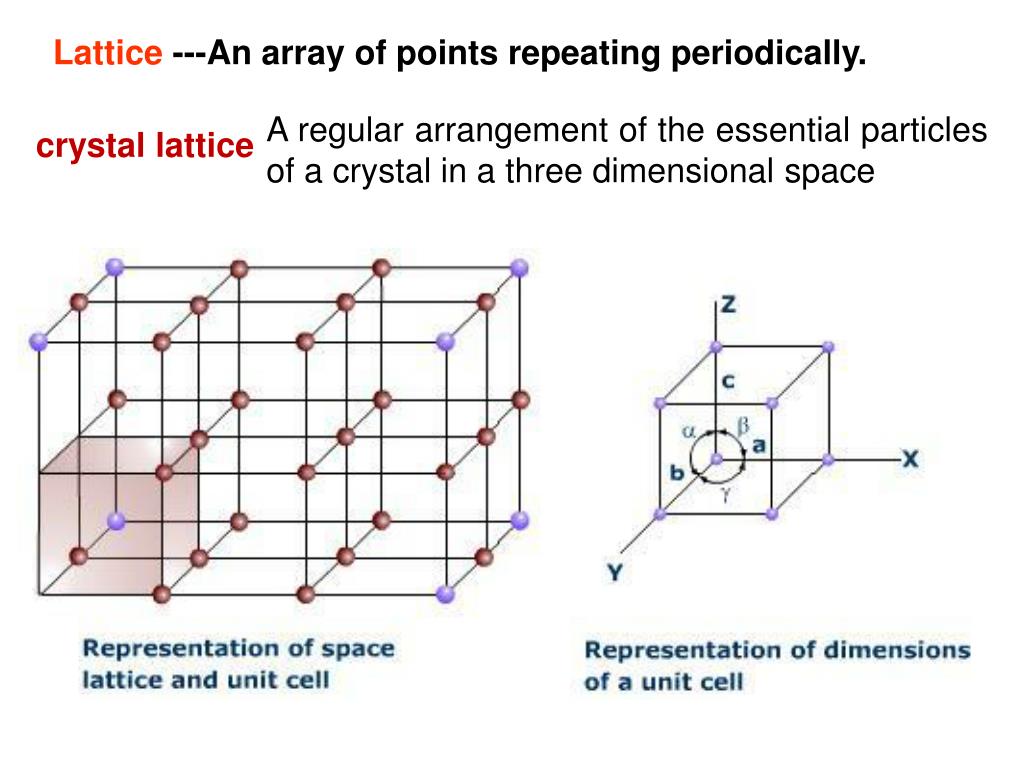
Also, seems that there is not a text dedicated to definitive concept. However, in general, when a text book of Inorganic Chemistry is consulted via INDEX appearst he word: "structure", in the sequence "subgroup", after "super". I think that the concept of sublattice belongs to inorganic physical-chemistry.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
